Author: Jordi Ferreiro
March 2026
1. Risk of contamination in Pharmaceutical Water systems
Microbial contamination and the presence of endotoxins are detected through sampling at the system’s Points of Use (POU). Applicable pharmacopeias establish acceptable microbiological limits (CFU/mL or CFU/100 mL, depending on the contaminant and water type).
For example, WFI: total count < 10 CFU/100 mL.
The verification and systematic monitoring of these critical parameters is mainly carried out through representative sampling at critical points and subsequent laboratory analysis, following pharmacopeial and regulatory criteria.
2. Sampling as a quality assurance concept
Proper sampling is essential to demonstrate compliance with the system’s microbiological requirements. It is complemented by continuous monitoring instrumentation, such as TOC and conductivity, which provide additional information on system performance.
Sampling strategy
- Availability of periodic sampling points at the outlet of critical equipment.
- Definition of appropriate operating ranges and levels, differentiating them from instrument design ranges.
These data allow the establishment of a critical quality control strategy for system qualification and operation:
- Alert and action levels
- Design, normal operating, and acceptable operating ranges
A system must meet design conditions and operate within the validated acceptable range to comply with GMP.
3. Continuous monitoring systems
According to EU GMP Annex 1, WFI systems must incorporate continuous monitoring (TOC and conductivity), which may be more representative of overall system performance than discrete sampling.
Limitations of conventional microbiological sampling
- Long time to obtain results (minimum 72 h)
- Detection limited to culturable microorganisms
- Susceptible to human error in CFU counting
- Provides estimated and partial values
- Inability to detect viable but non-culturable (VBNC) cells
- Risk of false positives due to external contamination
- High costs associated with deviation investigations and operational downtime
Continuous monitoring alternatives
There are currently systems based on bacterial fluorescence detection or cellular DNA staining that allow real-time estimation of microbial load in water, as a complementary solution to overcome or reduce the limitations of conventional methods.
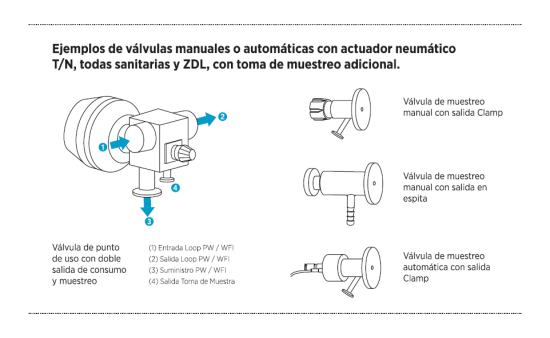
4. Recommended types of sampling valves
All system valves must be sanitary, diaphragm-type, and without dead legs (ZDL: Zero Dead Leg). This includes point-of-use valves and sampling valves, which must also have adjustable drainage.
Operational recommendations:
- Periodic opening of valves to minimize contamination risks
- Avoid using non-sanitary ball or seat valves
- For hose connections, ensure storage conditions allow complete drainage
Examples: manual or automatic valves with pneumatic T/N actuators, all sanitary and ZDL, with additional sampling ports.
5. Sampling guidelines and procedures to avoid false positives
The incorporation of sampling valves in Pharmaceutical Water systems is a key design element. However, it is even more critical that sampling operations are carried out rigorously and in a standardized manner to ensure representative samples and minimize microbiological false positives.
A detailed and validated Sampling SOP optimizes efficiency and reduces risks.
Main guidelines:
Alignment with user sampling procedures
During the design phase, it is highly recommended to review the sampling procedures used by the end user. This allows system design adjustments and identification of improvement opportunities based on installation type, accessibility, and automation level of valves and POUs.
Proper location of sampling valves
Sampling valves should be installed in positions that allow full opening and pre-flushing. Poor accessibility may increase the risk of external contamination and false positives.
Cleaning and disinfection prior to sampling
The procedure must define a thorough cleaning and disinfection process of the valve before sampling. Alcohol is commonly used, although the method should be agreed with the user according to their protocols and approved disinfectants. Proper disinfection can reduce microbiological excursions by up to 90%.
Pre-flushing before sampling
After disinfection, the sampling point should be flushed for at least 30 seconds at minimum flow to ensure the sample is representative of circulating water and not stagnant water in the valve or external contamination.
The recommended minimum flow corresponds to an outlet velocity > 2.44 m/s, equivalent to the following flow rates for typical ASME BPE outlet sizes:
- DN 1”: 3,375 L/h
- DN ¾”: 1,710 L/h
- DN ½”: 610 L/h
Trend-based monitoring and alert/action levels
The procedure should include a routine monitoring program with alert and action levels based on historical trends for each installation to detect sampling-related microbiological excursions early.
Inspection and periodic maintenance
Sampling valves must undergo regular inspection and maintenance to prevent dead legs, biofilm formation, and to maintain microbiological integrity.
6. Sanitization guidelines in pharmaceutical water systems
General recommendations
In addition to sampling programs, it is essential to define an appropriate sanitization strategy to both prevent contamination events and respond effectively when detected.
Without going into full technical detail, the following general recommendations apply:
Removal of non-sanitary components
If non-sanitary valves or components are detected, they should be replaced, followed by system sanitization and reinforced monitoring.
Prevention of biofilm formation
The main objective of sanitization is to prevent biofilm formation. Once developed, biofilm is extremely difficult to remove, even with strong disinfectants, so strategies must always be preventive.
Common types of sanitization
Sanitization methods in pharmaceutical water systems can generally be ranked as follows:
- Thermal sanitization (temperatures ≥ 65 °C)
- Ozone sanitization, mainly used in cold WFI systems stored below 18 °C
- Chemical sanitization, less recommended due to downtime and residue removal requirements
Thermal sanitization: influence of temperature and time
Thermal sanitization is considered the most robust method due to its high microbiological effectiveness and because it does not introduce additional substances into the water.
However, higher energy consumption has led in some cases to cold generation systems using ozone.
Its effectiveness depends on the combination of temperature and contact time.
Pharmacopeias and regulatory bodies generally recommend sanitization temperatures above 65 °C, as referenced in:
- EudraLex – Annex 1: Manufacture of Sterile Medicinal Products
- ISPE Baseline Guide for Water and Steam Systems (2019)
Recommended thermal sanitization times
Based on available studies:
- 4 hours at 65–70 °C
- 2 hours at 70–80 °C
- Minimum 1 hour at 80–95 °C
Intermittent flushing at valves is recommended during the process to ensure full surface contact.
Continuous vs. intermittent sanitization
Continuous thermal sanitization generally provides better microbiological control, maintaining less favorable conditions for microbial growth and avoiding residues.
Intermittent sanitization may be acceptable if:
- High frequency is maintained based on system trends
- It is adapted to plant operation (e.g., nights or weekends)
- A preventive approach is used (not only reactive to contamination events)
7. Design recommendations to minimize contamination risks and facilitate sampling
From a microbiological control perspective:
7.1 Selection of sampling valves and POUs
Define, together with the end user, the type of sampling and point-of-use valves according to the sampling procedure.
7.2 Identification of critical sampling points
Identify and agree on critical system points where sampling should be implemented.
7.3 Accessibility of sampling points
If access is limited, solutions should be implemented:
- Ensure safe access to elevated locations
- Manage low points (siphons) with proper drainage valves
- Use off-line sections with automated valves and accessible sampling points if relocation is not possible
7.4 General component recommendations
Use exclusively:
- Fully sanitary, zero dead leg (ZDL) valves
- Additional sampling/purge outlets in automatic POUs or off-line sections when direct sampling is not feasible, unless otherwise justified by risk assessment
